Differential Scanning Calorimetry Testing (DSC Analysis)

Microtrace utilizes differential scanning calorimetry (DSC) analysis as one tool for sample comparison, characterization, and identification. DSC measures the heat capacity of a sample and a reference over a programmed increasing and decreasing range of temperatures. Comparison of the sample and reference data permits identification and quantification of endothermic or exothermic processes occurring within the sample over specific temperature ranges.
A sample may absorb or release additional heat at particular temperatures for several reasons. Most commonly, as the sample hits a phase transition (e.g., solid to liquid), more or less heat must flow into the sample to maintain its temperature relative to the reference. DSC allows for accurate measurement and graphical representation (thermogram) of the amount of heat absorbed or released during such transitions. Anomalous peaks in these thermograms may indicate the presence of impurities in a sample. When a peak appears in a thermogram of the first, but not the second heating of the same sample, this indicates the presence from thermal “artifacts.” In these cases, the particular peaks reflect the sample’s thermal or mechanical history.
DSC Analysis of Polymers
Differential Scanning Calorimetry is a particularly useful tool to analyze polymers. DSC can reveal several important thermal transitions in these materials. Identification of a polymer’s melting, crystallization, and glass transition temperatures can help us characterize and compare such materials. Our experts can compare this data with well-cataloged standard references that exist for most polymers. For example, the thermograph below shows the results of a second heating applied to a sample. The two distinct thermal transition points (labeled) aided in its identification as Nylon 6.
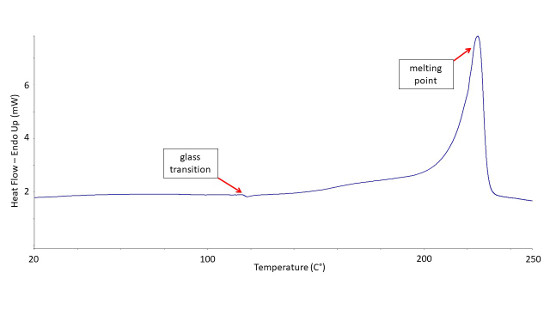
Simplified DSC Thermograph for Nylon 6.
In addition to identification, our experts can utilize DSC analysis for many other purposes. We can compare the crystallization and melting points from the first heating with standard references to determine the crystalline content of a known polymer. Identification of boiling points can permit the detection of individual plasticisers in a sample. We can also use Oxidative Onset Temperature (OOT) to analyze the thermal degradation of polymers. Because DSC measures numerous characteristic properties of a sample, its applications are vast.
DSC Specifications

Perkin Elmer DSC 8000 with autosampler
Microtrace uses a Perkin Elmer Differential Scanning Calorimeter (DSC 8000). This instrument uses a double furnace to simultaneously expose the sample and reference to temperatures ranging from -80⁰C to 750⁰C. The DSC 8000 is capable of heating and cooling samples at an wide range of rates. It can heat samples as slowly as 0.01⁰C per minute to as quickly as 300⁰C per minute; cooling rates range from 0.01⁰C to 150⁰C per minute. Microtrace’s DSC 8000 is also equipped with an autosampler. It automatically loads samples into the furnace and runs programmed thermal analysis experiments. Typical samples range from 3 to 10 mg.
Other Related and Complementary Techniques to DSC at Microtrace
Gas Chromatography- Mass Spectrometry (GC-MS)
DSC Standardized Methods
ASTM (American Society for Testing and Materials) has created several standardized guides for DCS analysis:
- E1356 – 08 (2014) “Standard Test Method for Assignment of the Glass Transition Temperatures by Differential Scanning Calorimetry.”
- E2009- 08 (2014) “Standard Test Methods for Oxidation Onset Temperature of Hydrocarbons by Differential Scanning Calorimetry.”
- D3418 – 15 (2015) “Standard Test Method for Transition Temperatures and Enthalpies of Fusion and Crystallization of Polymers by Differential Scanning Calorimetry.”
- D3895- 19 “Standard Test Method for Oxidative-Induction Time of Polyolefins by Differential Scanning Calorimetry.
- D7426- 08 “Standard Test Method for Assignment of the DSC Procedure for Determining Tg of a Polymer or an Elastomeric Compound.”
ISO (International Organization for Standardization) also has a standardized guide for DSC analysis. ISO 11357-2 (2013) “Differential Scanning Calorimetry and Determination of Glass Transition Temperature and Glass Transition Step Height.”
How May We Help You?
Contact usto discuss your project in more detail.







